Contaminated bath water skews refractive index results. New technology can accurately measure aqueous cleaning agent concentration.
Numerous critical process parameters must be monitored and controlled to efficiently and effectively yield the expected cleaning results for a qualified cleaning process. For batch or inline spray-in-air cleaners, these include items such as wash temperature, spray bar configuration, nozzle selection, spray pressure, exposure time and cleaning agent concentration. The latter is the most difficult to monitor and subsequently control for as it is progressively compromised, as it becomes loaded with contaminants and in particular, flux residue. For many years, the standard method for assessing wash bath concentration has been refractive index. Its primary instrument, the refractometer, is inexpensive and easy to use, and the process is fast. This is certainly an effective method for measuring a fresh bath, and when combined with pH measurement, an operator can assess the organic and alkaline levels of ingredients in the wash solution. One must note that the refractive index of a medium is a measure of the speed and direction of light passing through the medium, and this is ideal for a pure solution (Figure 1). For a fresh cleaning agent, there is a linear relationship between the refractive index value and cleaning agent concentration.
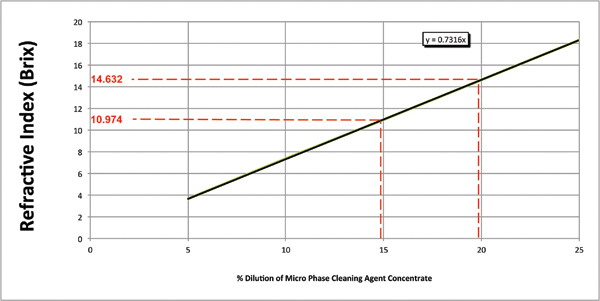
Figure 1. Concentration by refractive index.
In the world of aqueous cleaning, wash baths become contaminated with flux residues over time. Flux residues affect the speed and direction of light as it passes through the sample medium, thereby introducing unpredictable measurement errors.1 This means the measured concentration of the cleaning agent may be significantly over- or understated. For example, the targeted concentration may be 10%. The operator checks the wash bath concentration and discerns through refractive index a concentration of 20%. Thus, DI-water is added to reduce the concentration to the specified level. The actual wash bath concentration may well have been at 10%, and since an erroneous reading was obtained, and the wash bath was diluted, the actual concentration is significantly lower than the desired amount. The result: an out-of-spec process to worsen and therefore unknowingly yield assemblies outside the quality specification. Thus, identifying an improved test method to easily and accurately determine wash bath concentration is critical to an efficient and effectively managed aqueous cleaning process.
How does one accurately determine the concentration of chemistry, or an aqueous-based cleaning solution that comprises flux residues, as well as a wide variety of SMT-related residues that are entrained in a cleaning system wash bath?
For cleaning agent manufacturers that develop and blend their solutions, gas chromatography (GC) is an ideal test method for fresh or uncontaminated product. In this case, all constituents are known to the manufacturer and can be easily identified. When trying to examine contaminated samples with unknown constituents, having to select appropriate columns ahead of the analysis is difficult at best. Complications arising from the presence of unknown components are common to virtually all chemical analysis techniques, including chromatography. Thus, it is necessary to rely on experienced analysts making sensible accommodations for the presence of unknown species based on the fact that major contaminants can be reasonably guessed using flux formulations. It is also necessary to refrain from expecting the same quantitative accuracy, as in uncontaminated samples. With such caveats, GC remains a useful, yet non-practical technique for analyzing contaminated samples.
The chemical situation is further complicated in a mathematical sense by the fact that the total number and type of species within a contaminated wash bath is unknown. If it were somehow known that N different species were present in a sample, selecting a battery of analysis techniques that can give N different analysis values would provide a deterministic signature for the chemical profile. Thus, in such a multi-species environment, the practical approach to characterization is to choose sensing techniques based on their selectivity; that is, one chooses a sensing technique that returns a large signal for the species of interest, and a small or no signal for other species.
Trying to bridge the gap between the inaccuracy of refractive index measurements, when used for contaminated wash baths and the inherent difficulty of analyzing unknown and non-volatile species through the use of GC, has fueled the drive to develop alternate technologies. One approach to maximizing selectivity is to target the chemical behavior of the known components of the cleaning agent. This approach enables the chemists who develop the cleaning agents to choose from a very large universe of possible chemical behaviors and reactions to precisely target the desired species. Therefore, the next step in the evolution of concentration measurement tools was the development of the chemically targeted phase separation analysis technique.
One such commercially available technique is the Zestron Bath Analyzer. This was designed as a targeted physical reaction for the selected cleaning agents. During its development and beta testing, the results obtained using this technique were compared directly against those obtained through GC analysis for both a fresh cleaning agent (Figure 2), as well as a flux contaminated bath at a customer location (Figure 3). In each case, a micro phase cleaning agent was analyzed.
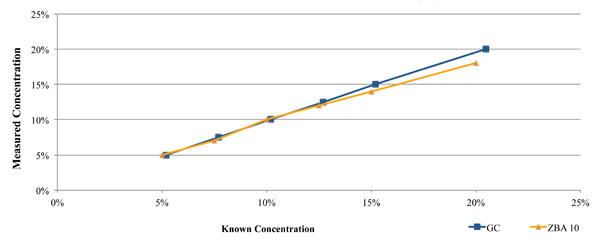
Figure 2. Results of bath analysis compared to fresh micro phase cleaning agent using known concentration.
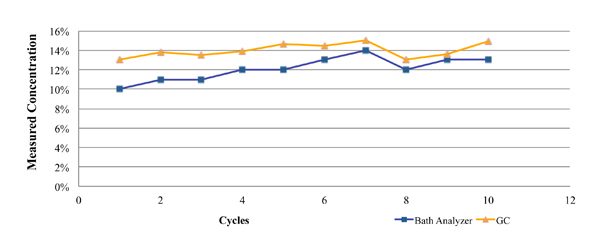
Figure 3. Results of bath analysis compared to fresh micro phase cleaning agent using known concentration.
As seen in Figure 2, the concentration measurement results using the GC and novel techniques accurately indicate concentration when compared directly against the known concentration of fresh aqueous engineered cleaning agent.
Figure 3 represents GC and novel technique data from a customer location. These data were developed through the analysis of partially loaded bath samples collected from a batch cleaning application over a four-week period. As noted in the graph, the concentration as measured by the novel technique closely mirrored that of the GC analysis, with a maximum difference of 3%.
With the novel technique, a wash bath sample may be analyzed in real time. On the basis of this analysis, concentrate or DI-water could be added as required to maintain the desired wash bath concentration. Additionally, when analyzing alkaline products, a color reaction indicates if the alkalinity of the wash solution is satisfactory, which is an indication of wash bath life. The novel technique is a manual process whereby the operator must extract a well-mixed wash sample, typically from the spray bar, monitor the process daily and add concentrate and/or DI-water as required.
Figure 4 represents data from a company that was using an inline aqueous cleaner and micro phase cleaning agent. In this case, there was a wide concentration process window ranging from 12.5% to 20%, with an average of 16.25%. The process was monitored over the course of several weeks by both refractive index and the novel technique, and the concentration manually corrected as required. As can be seen in the graph, it is clearly evident when the concentration drifted and when corrective action was taken – that is, concentrate added to bring the wash solution back into the targeted concentration range. Throughout the testing period, the concentration determined by refractive index continued to be overstated, reaching a maximum reading of nearly 38%. Had the operator reacted to this erroneous reading by adding DI-water, the wash bath concentration would have been significantly diluted, inhibiting cleaning effectiveness and potentially impacting the reliability of the electronic assemblies.
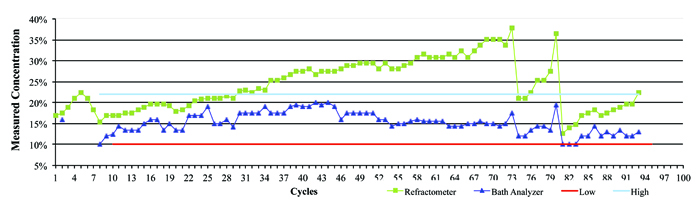
Figure 4. Field data showing refractive index vs. bath analysis.
Since 2009, the novel technique has been recommended as an alternative to refractometry for fast, accurate wash bath concentration measurement. However, the desire remained to develop a method that could indicate accurate wash bath concentration automatically, consistently and in real time.
The authors determined that the solution for assessing wash bath concentration resided in measuring liquid flow concentration “in line” as the wash solution is pumped to the spray bars, for this is the liquid that is in direct contact with the assembly surface during the cleaning process. Careful consideration was given to numerous available sensor options, including capacitance, acoustic and optical. However, identifying a technology and adapting it such that its output can be calibrated for an aqueous-based cleaning agent in the presence of flux residues, as well as other process contaminants, posed a significant challenge.
As noted, fresh wash bath concentration is an engineered aqueous-based cleaning agent, and thus all constituents and their relative proportions are known to the manufacturer. Given this fact, numerous concentration measurement technologies were considered by the authors and ultimately an acoustic method chosen as most appropriate for this application. This decision was made primarily due to its selectivity toward the desired cleaning agents under consideration. However, the authors understood that additional characteristic data of the flowstream were required to account for entrained contaminants. Here, we address the technological advancements made in adapting liquid flow sensor technology in order to develop a concentration monitoring process capable of accurately measuring wash bath concentration in the presence of flux loading.
Methodology
Numerous flux types and engineered cleaning agents are used in electronics manufacturing. To focus this study, the authors decided to conduct all tests using one of the modern cleaning agent types – that is, micro phase technology, herein referred to as the cleaning agent, as well as one flux type. The selected flux type was mildly activated rosin flux (RMA). Given this flux scenario, rosin would be a significant contamination contributor within the wash bath. Thus, for all flow sensing technologies evaluated, a key requirement was minimal selectivity toward rosin.
As discussed, there are numerous methods to determine wash bath concentration. For fresh as well as flux-loaded wash baths, the authors had significant data from in-house tests and customer sites using GC and the novel techniques. Thus, it was decided that the flow sensor concentration data would be compared against GC and the novel technique as a point of reference for concentration accuracy throughout this study. All GC measurements were performed at a Zestron Technical Center.
An acoustic measurement technology was chosen as the most appropriate automated concentration monitoring technique for the aqueous-based cleaning agents. The selected device, called Flowsensor, included an inline flow sensor and digital controller for concentration and temperature display. Ultimately, two types of these novel sensors were evaluated: a handheld benchtop device for single point measurement and an inline device for continuous measurements. Both capture not only acoustic wave data, but also the signal amplitude and signal dampening effect over time. The results of each were found nearly identical.
Utilizing the novel sensors, data from various benchtop experiments as well as spray-in-air cleaner installations were collected. All initial testing was conducted with fresh cleaning agent mixed to known concentrations. Using an algorithmic calculation, the sensor data stream was characterized such that the raw novel sensor data could be converted to a concentration percentage. These data were compared against both GC and the novel technique concentration measurements to evaluate and confirm sensor accuracy.
Figure 5 represents the GC, novel technique and novel sensor data as compared to the known cleaning agent concentration. For the novel sensor measurements, the cleaning agent temperature was 60°C, whereas for refractometry and Bath Analyzer, measurements were taken at room temperature. For GC analysis, the standard temperature protocol was followed.
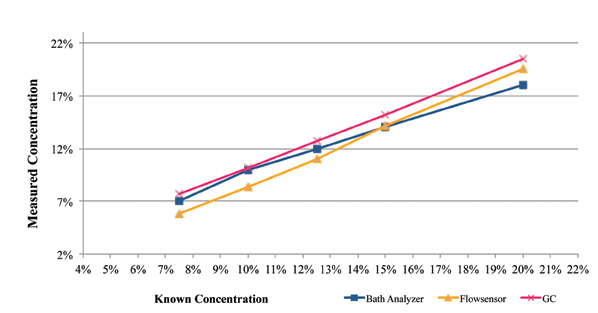
Figure 5. Cleaning agent concentration analysis.
As indicated by this data set, the GC and the novel measurement techniques accurately measured the concentration. The novel sensor measurement was within 1.6% of the known concentration at 10% percent concentration and above.
At this stage of product development, the novel sensor concentration variance was deemed acceptable. The next step was to consider the impact of flux residues on the novel sensor concentration measurements.
The process of analyzing the impact of flux residues was conducted in three stages. Since rosin is a significant component of RMA flux, the authors first examined its
impact on the novel sensor measurements. Following this assessment, the impact of flux residue was examined through controlled tests and, finally, wash bath analysis from beta site testing at a customer location.
A design of experiment was developed and executed in three phases:
Phase 1: Rosin load analysis, to evaluate the impact of rosin on concentration monitoring techniques, including the novel sensor and refractive index.
Phase 2: RMA flux load analysis, to evaluate the impact of various amounts of flux residue on various concentration monitoring techniques, including the novel sensor, bath analysis and refractive index. This test is to be conducted in the absence of any other SMT assembly contaminants.
Phase 3: Beta site test data. Employing the novel sensor, wash bath concentration field data were assessed by direct comparison to the bath analysis, refractive index and GC techniques.
Regarding cleaning agent temperature measurements, all novel sensor measurements were taken at 60°C; refractometry and bath analysis measurements were taken at room temperature, and the standard temperature protocol was followed for all GC analyses.
Main Research
Phase 1: Rosin load analysis. This analysis was conducted in a controlled environment whereby the volume of rosin and known concentration of the micro phase cleaning agent could be precisely measured, mixed and analyzed at a constant temperature.
To facilitate this evaluation, a custom engineered Automated Laboratory System (ALS) complete with the novel sensor (inline) was developed and employed. Initially, a 15% volumetric mixture of cleaning agent in DI-water was prepared. The liquid solution was dosed into the ALS with precise experimental accuracy. Then, measured quantities of rosin were added into this mixture to a maximum of 1.8%. In a typical inline cleaner with an 80-gal. wash tank, 1.8% rosin equates to 6 kg, a significant contamination load.
The rosin content was measured using refractive index and the novel sensor, with the measurement concentration values plotted in Figures 6 and 7, respectively. As indicated in Figure 6, the refractive index concentration measurements of the 15% prepared solution of the cleaning agent ranges from 14% to 28% at 1.8% rosin load. Thus, the refractive index measurement technique is extremely sensitive to rosin content. As indicated in Figure 7, the novel sensor concentration measurements of the 15% prepared solution of the cleaning agent ranges from 14.75% to 14.9%.
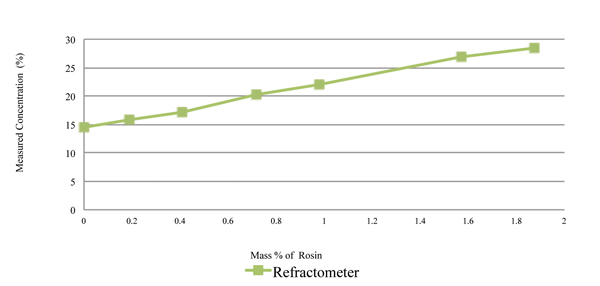
Figure 6. Rosin content measured using refractive index, with 15% cleaning agent and rosin.
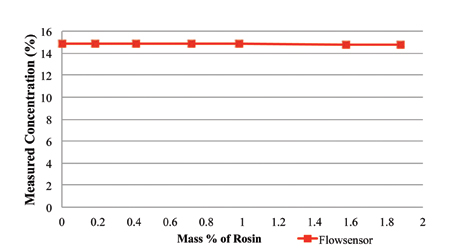
Figure 7. Rosin content measured using the novel sensor, with 15% cleaning agent and rosin.
Figure 8 is a combined graph detailing the impact of rosin on the cleaning agent concentration reading by refractometer and the novel sensor. Thus, acoustic measurement nearly ignores the rosin. It was concluded that rosin greatly impacts the accuracy of refractive index readings, yet has minimal impact on an acoustic measurement technique.
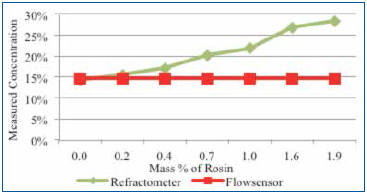
Figure 8. Novel sensor and refractometer results using 15% cleaning agent and rosin.
Phase 2: RMA flux load analysis. To evaluate the impact of flux load on the measurement methodologies, including the bath analysis, refractive index and the novel sensor, the evaluation was executed in two stages:
- Stage 1: Benchtop testing utilizing a 1000 mL beaker and a novel sensor (handheld).
- Stage 2: Installed novel sensor (inline) in the liquid flow line of a spray-in-air batch cleaner and conducted various wash cycles at different concentration levels.
For this test, high solid content RMA liquid flux was sourced. Since these tests were conducted using 1000 mL beakers as well as batch cleaners, concentrated liquid flux was used to minimize the overall flux volume required. To create the concentrated flux, liquid flux was evaporated such that the volume was reduced until it reached the known solid content.
For stage 1, two tests were conducted, Test A and Test B, each one using one liter fresh wash solution samples prepared to known concentrations. For each test, liquid flux with a solid content of 50% was concentrated through evaporation. It should be noted that in the authors’ experience, 3% flux load represents a worst case scenario. Other studies have found the same.2
Typically, one would expect a flux load average of 1.5%, particularly for an inline cleaning system. Generally, flux loading reaches an equilibrium point. As the wash solution is lost due to evaporation and drag out, the concentrated cleaning agent and/or DI-water is added, thereby minimizing the upper limit of flux accumulation.
Test A (benchtop tests): Concentrated flux was added to a 12% concentrate cleaning agent solution in 0.5% increments to a maximum of 3%. Following each flux addition, the concentration was measured by the novel sensor and bath analysis (Figure 9).
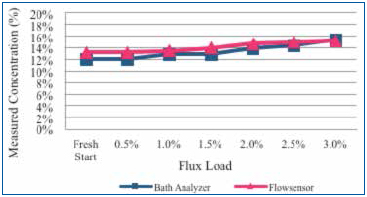
Figure 9. Measure of concentration using novel sensor and bath analysis method as flux influence increases from 0.5% to 3%.
Test B (batch cleaner tests): Concentrated cleaning agent at 7.5%, 10%, 12.5%, 15%, and 20% was prepared. In this case, each concentrated sample was loaded with flux to 3% by volume. The concentration of each flux loaded solution was measured using refractive index, GC and the novel sensor (Figure 10).
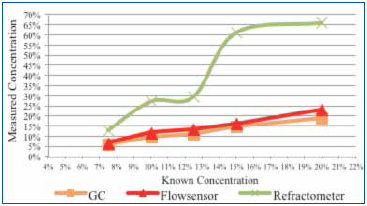
Figure 10. Concentration of each flux loaded solution measured using refractive index, GC and the novel sensor. Flux influence: 3%.
Results. For Test A, the initial wash bath concentration target was 12%. Fresh bath concentration readings were 12% and 13%, as indicated by the bath analysis and the novel sensor, respectively. As the flux concentration increased, the indicated wash bath concentration increased to 15%, as shown by both measurement methods. Note that the indicated concentration value for either measurement technique varied by approximately 1% at a flux load of 1.5%. In this experiment, the flux addition induced a maximum intrinsic error of 3% at a concentrated flux loading of 3%.
For Test B, five concentrations, each loaded with 3% flux by volume, were measured using GC, the novel sensor and refractive index. As expected, refractive index measurements increased exponentially as the cleaning agent concentration increased, greatly overstating the concentration. On average, GC understated the known concentration by 1%. The novel sensor measurements overstated concentration by a low of 0.77% (5.77% vs. 5%) to a high of 2.94% (22.94% vs. 20%).
For Stage 2, the novel sensor (inline) was installed in the wash pump discharge line of a spray-in-air batch cleaner. The process settings used for this test are detailed in Table 1.
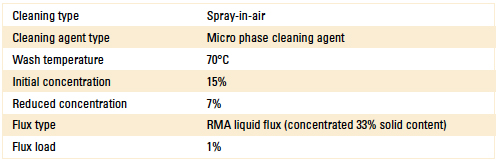
Table 1. Batch Cleaner Process Parameters
The wash tank was filled with fresh cleaning agent to an initial concentration of 15%, as verified by refractive index, bath analysis and the novel sensor. The cleaner basket remained empty, as this test was examining the effect of flux load on concentration measurement only. For this test, the liquid flux solid content was 33% and again concentrated through evaporation.
Following cycle start, flux was added, then diluted, and concentration re-measured employing refractive index and the bath analysis. The novel sensor continuously recorded wash bath concentration throughout the test.
Results. The initial wash bath concentration measured by refractive index and bath analysis was confirmed at 15% concentration.
Flux was added to a concentration of 1% by volume. During this wash cycle, the novel sensor measurements remained fairly constant throughout (Figure 11). At time t2, the bath analysis measurement remained accurate, yet the refractive index measured 22% (actual 14%). The wash bath was then diluted with DI-water. The novel sensor measured 7% as also indicated by the bath analysis, yet the refractive index measured 15%.
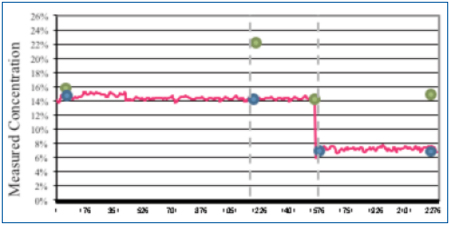
Figure 11. Concentration change analysis.
As expected, in the presence of flux residue, the refractive index reading overstated the wash bath concentration in both instances. At a 1% flux load, both the novel sensor and bath analysis very accurately indicated wash concentration, even as it was diluted by half.
Phase 3: Beta site test data. Based on the results of Phase 1 and 2 of the DoE, the authors proceeded with evaluating the novel sensor at a customer beta site. In this case, the customer had a qualified cleaning process for many years using an inline spray-in-air cleaner. Process parameters are detailed in Table 2.
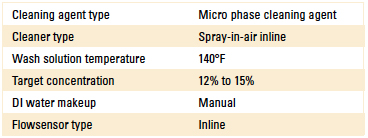
Table 2. Inline Cleaner Operating Parameters
Upon installing the sensor, a fresh wash bath was added and the concentration adjusted to the 12% target. As the customer monitored the concentration, DI-water was manually added using a dosing pump that was manually actuated as required. For this study, single point measurements were taken daily for four days following the initial startup using refractive index, the novel technique and novel sensor plotted in Figure 12.
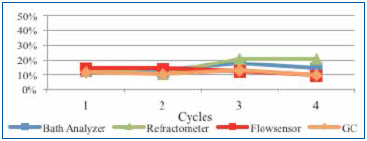
Figure 12. Beta site concentration readings.
The fresh wash bath concentration readings were very close between all measurement techniques at startup. However, as the production week progressed, concentrate and DI-water were added as required, as contaminants began to accumulate in the wash bath. Refractive index readings initially dropped and then increased, overstating concentration. The novel technique, GC and novel sensor measurements tracked consistently throughout the remainder of the week. Although this data stream is positive but limited, the customer is continuing to use and evaluate the measurement technology.
Conclusion
It is critical to determine the wash bath concentration, particularly as it becomes loaded with flux residue and other contaminants. Incorrect concentration measurements will lead to incorrect wash bath concentration adjustments. If the concentration is overstated, DI-water will be added, increasing dilution and potentially resulting in poor cleaning results and subsequently product reliability concerns. Alternatively, if the concentration is understated, concentrated cleaning agent will be added, increasing the wash bath concentration and the potential for material compatibility concerns.
The wash bath flux loading rate is certainly variable depending on the flux type used, solid content, board complexity, reflow profiles, and production volume to name a few. In this study, the flux analysis was limited to RMA. Rosin was shown to have a major impact on the accuracy of Refractive Index contributing to the overall inaccuracy of this measurement method. Alternatively, the rosin impact on the acoustic measurement device was minimal, thereby reducing its intrinsic measurement error.
GC can certainly be used for concentration measurement, even with contaminated samples. However, due to its cost and complexity, it is not feasible to use at the assembly plant level on an ongoing basis. The novel technique is a feasible alternative offering reasonable measurements – that is, within several percent of the target concentration as compared with GC analysis. This is a manual method and subject to human error.
Alternatively, the novel sensor as described herein, or the acoustic liquid flow measurement technique, is an inline device that continually and automatically monitors, records and displays the wash bath concentration in real time. Through the series of tests conducted and presented within this study with both fresh and RMA flux loaded cleaning agent, the authors have demonstrated that the novel sensor, once characterized for the specified cleaning agent and expected contaminants, accurately measures wash bath concentration in the presence of flux residue. For a wash bath solution loaded with 3% flux, the measurement overstated the target concentration in the range of 0.7% to 3%, depending on targeted concentration. The intrinsic error was less at a reduced flux load.
For this study, field data were limited yet consistent with the study test data and will continue to be collected.
Acknowledgments
Special thanks to Kester for providing the flux samples used for this study.
References
1. H.Wack, “Limitations of Refractive Index,” CIRCUITS ASSEMBLY, April 2009.
2. William T. Wright, “Managing Wash Lines and Controlling White Residue by Statistical Process Control,” Proceedings of SMTA International, September 2003.
Ed: This article was originally published in the proceedings of SMTA International, October 2013, and is reprinted here with permission of the authors.
Umut Tosun is an application technology manager, and Axel Vargas is a sales engineer at Zestron (zestron.com); umut.tosun@zestronusa.com. Bryan Kim, Ph.D., is director of engineering at Pressure Products (pressureproducts.com); bhkim@pressureproducts.com.
A systematic approach to nonconventional methods of encapsulant removal.
An unfortunate reality is that the circuits we assemble do not always perform as expected. Failure analysis is frequently needed to find root causes. In such cases, it is common to encounter components encapsulated with materials that must be removed to permit inspection. While “decapsulation” methods are well documented, with many guides available to help with products and “recipe” selection for concoctions to remove encapsulants (ASM Microelectronics Handbook, Dynaloy data sheets, etc.),1 the analyst will encounter materials in which standard methods, such as use of fuming nitric, sulfuric acids or commercial deprocessing products, fail to produce the desired outcome.
Most often, this poor outcome arises because the chemicals used to remove encapsulants also attack the materials of interest. For example, using nitric acid on sites composed of copper wire bonds results in almost no chance of the copper remaining unmolested for further analysis. This highlights the “magic bullet” misconception – belief that a chemical can be found that selectively attacks only the materials wanted, while not altering materials of interest.

Figure 1. Overall resistor construction revealed by x-ray imaging.
Methods such as breaking parts simply by fracturing or cleaving, burning away materials in a furnace, use of unconventional chemicals, or mechanical methods can often overcome standard methodology limitations and permit successful removal of encapsulants, while maintaining the integrity of the materials of interest. A systematic approach to such challenges and examples in which the Raytheon Failure Analysis Lab in McKinney, TX, addressed some of these are highlighted here.
In all things, it is generally a good idea to think about how to proceed before actually undertaking a task. In the case of decapsulation, several fundamental questions should be asked:
- What is the composition of the materials to be removed?
- What is the composition of the materials that are expected to be revealed?
- What are the properties of the materials?
- How will the materials respond to the decapsulation process?
The answers are crucial with regard to choosing the proper methods to remove the encapsulation while preserving the materials of interest. Labs that perform such procedures on a regular basis are certainly aware of such issues and likely have common procedures in place. A typical protocol might follow a procedure such as:
- Identify material compositions from data sheets or analysis in the lab. (FTIR spectroscopy is a quick method for determining most material classes of interest.)
- Consult a materials chart that provides guidelines as to proper procedures. For example, many suppliers of decapsulating agents provide charts that indicate which of their products to use for each material type.
- Follow proper procedures, with safety a key concern, and monitor the components at regular intervals to achieve the desired penetration, while maintaining the exposed materials’ integrity.
As mentioned, such procedures are typical and apply to most applications. However, the analyst also encounters examples in which standard procedures will not work and creativity is needed in planning the proper exposure route. The remainder of this article will focus on a few such examples faced by our lab and highlight the rationale behind these often unconventional approaches.

Figure 2. No fractures in the element or issues related to the end cap weld joint could be resolved.
Why Not Burn It Off?
It may seem counterintuitive that burning away material is an acceptable approach; after all, such a method is aggressive, seemingly difficult to control, and not very selective. However, in some cases, in which the goal is the burning away of combustible materials to reveal noncombustibles such as metals, the approach can overcome limitations of other more traditional techniques. For example, if the purpose of the decapsulation is to reveal fine metal features such as wires of a resistor, small bond wires, and others, the use of strong acids such as nitric often results in dissolution, or at least attack, of the metals of interest.
In the following example, multiple wire-wound resistors were failing in an open condition once installed on an assembly and exposed to environmental testing. X-ray analysis could not resolve an open condition in the nickel-chromium element. Therefore, a method was devised to selectively remove the encapsulant without affecting the fragile element, thus permitting further detailed inspection.
The package design was such that the wire element was wound over an alumina core with a heavily glass-filled encapsulant. Such a design seemed ideal for furnace removal of the encapsulant, as the materials to be revealed are capable of withstanding exposure to 500˚ to 600˚C and flames resulting from the burning of the epoxy. In this case, the leading concern was to avoid mechanically disturbing the delicate wires during the process. To ensure the approach was reasonable, a known good component was subjected to the proposed methods and found to pass electrically on decapsulation, an indication that the method did not damage the element.
The method was composed of the following steps:
- Packages were exposed to 500˚ to 600˚C in an appropriate furnace. Samples were monitored to determine complete combustion of the packaging epoxy (about 5 min).
- Packages were examined for completeness of the process. In such examples, very close control of the heat can overcome the problem encountered: the glass filler fused, forming a secondary layer that needed to be removed. Ideally, if the combustion heat is maintained at a sufficient temperature to burn away the polymer but not fuse the glass, the filler can be gently, mechanically removed. Unfortunately, in many cases, such as this example, the glass forms a secondary material that must be removed before the inner package surfaces are revealed.
- The fused glass was digested using hydrofluoric acid. After burning away of the epoxy, the package was placed in concentrated hydrofluoric acid with stirring, and monitored for complete removal of the glass. This is a weak acid that is very selective for glasses and very mild to most all metals. (There are exceptions.)
Figure 3 shows that the resistive element appears to be disturbed in areas adjacent to the fracture site indicated; this is due to a lack of tension in the wires at this region (due to the fracture) coupled with gentle agitation of the wires in the hydrofluoric acid. Regardless, the fracture site was easily identified and characterized.

Figure 3. Wire revealed on alumina core.
Figure 4 reveals features that indicate the wire was corrosively attacked. Additional failing samples were evaluated using the same deprocessing methods, and similar features were identified at the fracture locations. These were always in close proximity to the end cap terminations; this suggested that a contaminant may have “wicked” in during processing or environmental exposure. Although this deprocessing technique was useful in identifying the failure mode (corrosive attack), root cause had not been established, and any possible chemical agents that may have contributed to the corrosion were removed by the procedure.

Figure 4. SEM figure of Nichrome wire at fracture site.
In addition to the example cited, we have successfully deprocessed several parts using the furnace approach at such times when it is applicable. For example, in another case, very small aluminum wires encapsulated in a soft urethane coating were not revealed by x-ray examination. Burning away the potting in the furnace allowed us to successfully characterize the wires.
Mechanical Methodology to Minimize Chemical Exposure
Purely mechanical deprocessing methods were employed to test the chemical “wicking” hypothesis presented in the previous example. In such a process, no chemicals can be used that may contaminate the device or even remove suspect contamination. In this analysis, we had the luxury of multiple failing samples to utilize different complementary deprocessing methods. A subset of the failing devices was mechanically deprocessed using a cleaving tool to separate the molding and expose the element. Obviously, this technique was never intended to preserve the condition of the fragile element.
Isopropyl alcohol extracts of contaminants on the exposed element were captured and evaluated using GC/MS (gas chromatography/mass spectrometry) methods. The presence of glycerin was identified on all failing devices, a good target compound indicating the presence of water-soluble fluxes. Consultation with the supplier revealed that they did indeed use a water-soluble flux with these devices. As a result, a good device was similarly deprocessed and exposed to the flux with power applied to test the assumption that such exposure led to the open. Similar corrosive attack was noted. Corrective actions included changing to a more benign flux.

Figure 5. Corrosive attack resulting from the wire being exposed to water-soluble flux while powered.
This example highlights the use of hydrofluoric acid for digestion of glass, much like the previous example, but is used on a package that traditional methods were also employed. The semiconductor package displayed in Figure 6 contains stacked die and is configured with an interposer board. Upon visual inspection, the molding compound appeared to have a shiny smooth surface. (FTIR analysis showed this to be a heavily glass-filled epoxy.) To decapsulate, the package was submitted to the traditional methodology using fuming nitric acid. The molding compound displayed only limited removal using this approach. This was determined to result from the extremely high glass content in the encapsulant. (In such cases it is perhaps more useful to think of such materials as glass with polymer binder than a glass-filled polymer.) While the nitric acid was effective at removing the polymer, the glass at the outermost exposed layer concentrated, forming a barrier unaffected by nitric acid. It was decided a multi-step process alternating between nitric and hydrofluoric acids would address this issue. After the nitric acid ceased to be effective, 49% hydrofluoric acid was applied to the top layer for a minute or so. After this, the hydrofluoric acid was rinsed away and nitric acid was reapplied. This sequence was repeated three times, and decapsulation was successful.

Figure 6. A heavily glass-filled epoxy encapsulant.
The Problem with Silicones
Silicones are ubiquitous materials used in a multitude of applications, including coatings, underfills, thermal transfer compounds, adhesives, and others. It is desirable in many instances that such materials be removed from components during deprocessing. One of the most common methods is to soak these materials in solvents such as xylenes or hexanes, swelling and softening them for ultimate mechanical removal. This presents obvious issues when deprocessing delicate assemblies, such as mechanically destroying fine features during the removal process. Also, while a few silicone removal products are available from commercial sources, these rely on corrosive additives that digest the silicone rather than simply dissolving them. Such aggressive materials may compromise sensitive materials, and swelling often still occurs.
While we would like to claim credit for the following solution (we cannot), we have found scarce reference to a method reported by Sachdev.2 While the focus of the referenced article is silicone adhesives, we have found it equally applicable to all silicones we have tested, including condensation, and addition cure materials, including thermal transfer media, pottings, and “RTVs.”
We found that application of a solution of 1% tertabutylammonium fluoride (TBAF) in propylene glycol methyl ether acetate (w/v), as described by Sachdev, heated from 50˚ to 90˚C with gentle stirring works exceptionally well to remove silicones and not damage sensitive materials. (Higher heat and solutions with greater concentrations of TBAF proved to accelerate the digestions.)
Figure 7. Bond wire prior to silicone removal.
In the following example, a silicone potting covered very fine bond wires of a diode in a hermetic package. The goal was to examine the diodes and perform a bond-pull on the wires to document possible degradation modes after highly accelerated life testing (HALT). The typical methods of removing the silicone invariably caused dimensional changes in the silicone, which imparted an undesirable stress on the wires.
The new method was tried on a few test samples, and the results were excellent. No damage or deformation was observed. Bond-pull results compared favorably with the devices under test.
Another example of this technique that demonstrates its value is the failure investigation of a complex product with multiple layers of material. The failure scenario suggested the likelihood of broken bond wires. A partial view of the component is shown in microsection (Figure 8). The materials in this view are labeled, and the objective was to expose the wires for evidence of fracturing somewhere along the wire or at the bonds. Sectioning in silicone to reveal fine features cannot achieve this objective. Using traditional methods, once the silicone is exposed, the approach leads to swelling, cracking or other dimensional changes that could induce the very same damage in the bond wires that we are attempting to uncover.
Figure 8. Cross-sectional view of device.
This technique was used to dissolve the silicone after the epoxy over-mold was removed. This was first tried on a known-good and new test sample. Results are shown in Figures 9 and 10. There was no deformation of the bond wires. Subsequent attempts on failed samples allowed us to confirm fine heel fractures in the bond wires. Finally, Figure 11 shows how the method permitted fine details of the exposed bond wires to be recorded.

Figure 9. The silicone-encapsulated wires are visible after the epoxy was removed.

Figure 10. Wires shown after silicone removal. No deformation or damage was detected on the test samples, and heel fractures were found on the failed samples.
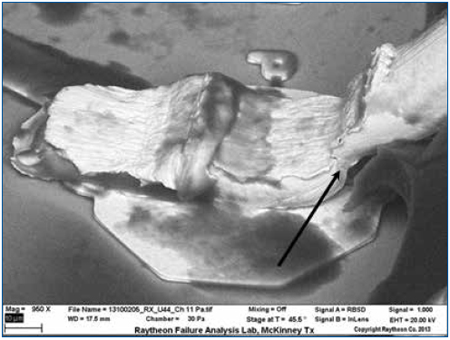
Figure 11. SEM of the wire bond shows that the silicone encapsulation has been sufficiently removed to permit inspection of fine details. Crack initiation is visible in the neck-down region (heel) of the wire.
Standard decapsulation methods are not always applicable, and the analyst must devise approaches to circumvent their limitations. While certainly not exhaustive, a few examples that provided unique challenges to our lab were highlighted. These were selected to show that, at times, unconventional solutions to such problems, when devised with materials variables in mind, can often prove quite effective.
References
1. The SAE G19A Test Laboratory Standards Development Committee will soon be releasing a decapsulation standard. Also, a web search of the terms “decapsulation of electronic components” will result in many additional references.
2. Krishna G. Sachdev, Ph.D., “Removing Cured Silicone Adhesive from Electronic Components," Solid State Technology, October 2010.
W. John Wolfgong, Ph.D., is a chemist; Jana Julien is a failure analyst; Jason Wheeler is a failure analysis engineer, and Joe Colangelo is a principal failure analysis engineer at Raytheon Space and Airborne Systems, Component Engineering Department (raytheon.com); wolfgong@raytheon.com.
Can improved flux coatings on solder preforms reduce voiding?
Void reduction is a critical challenge in electronics assembly, especially for bottom terminated components (BTC) such as QFNs (quad flatpacks no-leads). Excessive voiding inhibits heat transfer between the thermal pad on the QFN and the PWB pad. Flux-coated solder preforms have been shown to significantly reduce voiding.1 Here, the investigators studied the effects of flux coating improvements on voiding.
Sample preparation and experimental procedure. Eutectic SnPb or SAC 305 solder preforms were placed between two ENIG coated 1.25" x 0.375" x 0.008" copper-oxidized and non-oxidized substrates and reflowed in air (Figure 1). To ensure statistical confidence in the results, 30 samples were evaluated at each experimental condition. Half the experiments were performed with a typical flux coating (A) and the other half with a new flux (B). No mechanical pressure was used on the substrates during the ramp-to-peak reflow processes. A statistical comparison of the voiding performance of the two fluxes was then performed.

Figure 1. In the experiment, two ENIG-coated copper substrates were reflowed with flux-coated solder preforms. Half the copper substrates were oxidized.
Flux B is a halogen-free flux in 1% formulations.
The typical flux coating formulation (A), also at 1% concentration, produces inconsistencies and gaps in the flux coating, as seen in Figure 2. Gaps and inconsistencies often result in more voiding.

Figure 2. Gaps and inconsistencies in Flux A (left) will often result in more voids compared to Flux B.
All samples were processed in an air reflow, ramp-to-peak profile. Data were collected to measure the void content after reflow using x-ray imaging
(Figure 3).
Figure 3. Flux voiding was measured from the x-ray images. Note the much lower voiding level for Flux B.
Data Analysis
SAC 305 non-oxidized substrates. All experiments were performed with 1% flux formulations. A boxplot of voiding data for the non-oxidized SAC 305 substrates for both flux types is shown in Figure 4. The mean voiding value for the substrates treated with Flux B was 1.39%, whereas the value for substrates treated with Flux A was 2.79%. Performing a two sample t-test analysis indicated that Flux B produced fewer voids than Flux A with a statistical confidence of >99.99%.
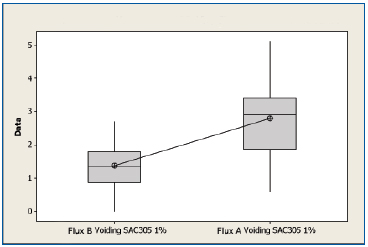
Figure 4. Boxplot of voiding data for non-oxidized SAC 305 substrates coated with Flux A and Flux B. Statistical confidence: >99.99%.
SAC 305 oxidized substrates. A boxplot of the voiding data for the oxidized SAC 305 substrates for Flux A and Flux B is shown in Figure 5. The mean value for the substrates treated with Flux B was 1.79%, whereas the mean value for substrates treated with Flux A was 2.71%. Performing a two sample t-test analysis indicated that Flux B produced fewer voids than Flux A with a statistical confidence of >99.9%.
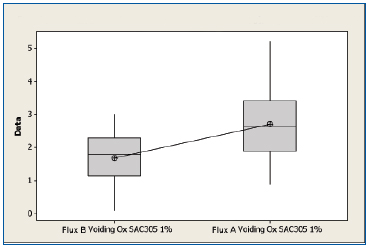
Figure 5. Boxplot of voiding data for oxidized SAC 305 substrates coated with Flux A and Flux B. Statistical confidence: >99.9%.
SnPb non-oxidized. A boxplot of voiding data for the non-oxidized SnPb substrates for both fluxes is shown in Figure 6. The mean voiding value for the substrates treated with Flux B were 0.327%, whereas the substrates treated with Flux A was 1.89%. Performing a two sample t-test analysis indicated that Flux B produces fewer voids than Flux A with a statistical confidence of >99.99%.
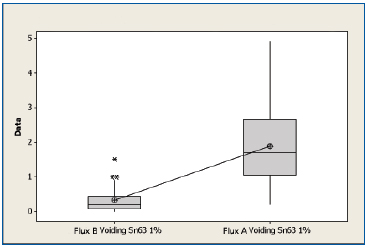
Figure 6. Boxplot of voiding data for non-oxidized SnPb substrates coated with Flux A and Flux B. Statistical confidence: >99.99%.
SnPb oxidized. A boxplot of voiding data for the non-oxidized SnPb substrates for both fluxes is shown in Figure 7. The mean voiding value for the substrates treated with Flux B was 0.197%, whereas the value of the substrates treated with Flux A was 2.29%. Performing a two sample t-test analysis indicated that Flux B produces fewer voids than Flux A with a statistical confidence of >99.99%.
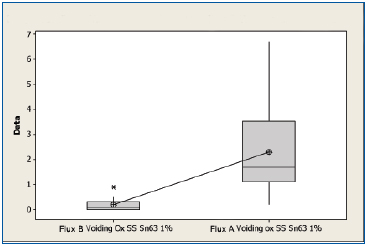
Figure 7. Boxplot of voiding data for oxidized SnPb substrates coated with Flux A and Flux B. Statistical confidence: >99.99%.
Summary
Void data are summarized in Table 1. In all cases, there is a strong statistical confidence that Flux B performs better than Flux A. The new Flux B is halide-free and improves wetting. Flux B coats the solder preform more uniformly to further improve wetting. These features significantly reduce voiding when preforms are used with BTCs such as QFNs. Additionally, Flux B leaves less flux residue.
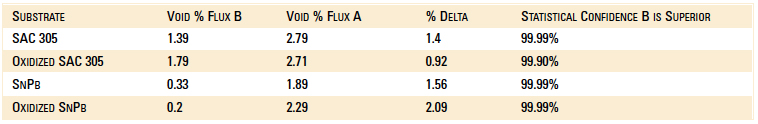
Table 1. Voiding Data Results
References
1. Seth Homer and Dr. Ronald C. Lasky, “Minimizing Voiding In QFN Packages Using Solder Preforms,” IPC Apex, February 2012.
Seth Homer is assistant product manager engineered solders, thermal materials and NanoFoil preforms at Indium (indium.com); shomer@indium.com. Dr. Ronald C. Lasky is senior technologist at Indium.
“DFRM” reveals potential manufacturing issues and the sample of processes that can feed those issues back for design review.
As detailed in CIRCUITS ASSEMBLY last year (“Robotics Assembly from Prototype to Production,” March 2013), robotics has received an increase of attention in the media thanks to overall advances in the technology.1 This article details some of the key areas of design for robotics manufacturing (DFRM), and provides recommendations for products transitioning from the design to production phase.
During the prototype or design verification build stage, the main goal is to prove out the bill of materials (BoM), assembly drawings and overall robotics design. This is the phase when manufacturing issues are documented and fed back for resolution before a product is released to production.
The following are examples of areas of focus during the prototype review:
Form, fit and function. Two validations exist during the form, fit and function stage. First is a design review of the build documentation, including design models, BoM and assembly drawings. Depending on tools available, the review could be electronically automated or manual, with the result a validation that the overall build documentation meets the intended design intent. The second validation is the actual assembly of the robot and how all the material fits together. Problems may occur with sheet metal, plastic material or wire routing interference issues. Wire routing may require additional review, given the opportunity of pinching, wires unplugging or other reliability concerns. If any of these issues occur, an engineering change order (ECO) may be required to the design model and assembly drawings.
Rework or modification to material may be required to make the prototype “fit” together. As with any modification, an increased attention should be made on the impact to the next-level assembly. The important takeaway of this rework is to incorporate a permanent correction into the next assembly build or product roadmap. Issues may also occur during revision changes of a dimension modification. The goal is to verify the fit, modify if necessary and address and incorporate all changes before the follow-up build or before the introduction-to-production phase.
During the design and build review, consider analysis tools such as PFMEA (Process, Failure, Mode, Effects and Analysis) or equivalent to assess areas of the process in which error proofing (Poka yoke) tools or methods can be used. The PFMEA review can look at materials, methods, process and equipment. Similar analysis can be completed during the initial design stage (DFMEA - Design, Failure, Mode, Effects and Analysis). These reviews assess the areas at risk, categorize and prioritize the risk, and determine what can be implemented to prevent the issue from occurring. Typically, this exercise is completed with a cross-functional team to ensure a variety of backgrounds, experiences and knowledge, which ensures a comprehensive assessment is performed.
Ease of manufacturing. Following the DfM process after the form, fit, function review, ease of manufacturing is assessed. Ease of manufacturing identifies ways to reduce product cycle time and improper quality. These ideas range from the simple to the complex. Examples include using captive washer screws or pre-applied adhesive verses hand-applying adhesive. Another example is having multiple wires or cables harnessed together for ease of routing and assembly.
In addition to changes in material, correct selection of equipment and tooling can greatly help the ease of manufacturing. If an assembly has many screws at the same operation and torque setting, an electric or automated screw driver may assist. An assessment may be required to justify the expense or return on investment of the purchase. Custom designed fixturing may be the most helpful of all ease-of-manufacturing recommendations. These fixtures may assist with product support, various assembly operations (such as press-fit), material handling and inspection. Figure 1 shows an example of a custom fixture, designed and manufactured at Benchmark Electronics NH Division for a medical robotics program. Some of the benefits of this custom fixture include reduced process cycle time and increased product quality through better material handling.

Figure 1. Custom designed turntable fixture for ease of manufacturing. (Courtesy Benchmark Electronics NH Division)
Quality control and test review. As discussed above, the initial design build is an important DfM review tool to ensure form, fit and function of a robotics assembly. Two additional important inputs result from quality control and testing of the subassemblies and completed robotics assembly. These two inputs will also be fed back to the DfM review as potential design change for future builds.
During quality control or visual inspection, among the items reviewed include adherence to internal and external criteria, industry standards and potential areas of concern for functionality of the subassemblies. Cosmetics issues will also be reviewed for the completed robot, usually focusing on external surfaces visible to the end-customer.
Depending on the end-application, various noise levels such as fans, motors and gears may be an area of DfM feedback.
The acceptable noise level for a robot in an industrial or military application may be quite different from one in a medical or office environment.
Figure 2 shows an example of a gear system. In cases of motors and gears, testing feedback through the subassembly and final robotics system test will impact DfM review. Testing will verify design, functionality and application with a complete load placed upon the assembly. Be aware of the noise and overall decibel level and consider the final customer application and environment.

Figure 2. Sample gear system. (Courtesy howstuffworks.com)
References
1. Scott Mazur, “Robotics Assembly from Prototype to Production,” CIRCUITS ASSEMBLY, March 2013.
Scott Mazur is manufacturing staff engineer at Benchmark Electronics (bench.com); scott.mazur@bench.com.
How Aqueous Technologies is leading the way toward clean assemblies.
It all started, fittingly, on a napkin.
Below the San Bernardino Mountains in Rancho Cucamonga, CA, you’ll find Aqueous Technologies, a cleaning company launched more than 20 years ago. The manufacturer of cleaning systems for electronics assemblies is the vision of Mike Konrad, president and CEO.
Konrad, described by colleagues as having “a special entrepreneurial spirit,” saw “water as the future” of cleaning boards when he started in a garage in Northern California. It was in that garage that Konrad designed his first cleaning machine, outlining it in pen on a napkin. That makeshift parchment now resides, in a frame, at the current company headquarters.
Konrad was at the time working for a soldering equipment manufacturer. It was the 1989 signing of the Montreal Protocol, which eventually banned CFCs, then the primary cleaning solvent for printed circuit boards, that inspired him to launch Aqueous Technologies. “There was considerable concern, even panic as there was no real (legitimate) alternative,” recalled Konrad in an interview.
“At that time, there were water-based cleaners, but they were little more than mildly converted dishwashers and were better at soliciting jokes than cleaning boards.” Still, the basic technology behind these water-based cleaners had promise.
Konrad suggested the development of a water-based alternative to solvent cleaners, but his employer wasn’t interested. Funding the startup himself, Konrad and a friend designed a water-based cleaning system, which they sold to his employer. Emboldened, he began developing designs for systems that would be more environmentally safe, including a zero-discharge cleaning system, which, he says, had no impact on the environment. That’s where his employer balked and Konrad walked.
“When I approached my employer with that idea, they were not impressed. Seeing no need, they rejected my idea. With necessity being the mother of invention, and rejection being the mother of motivation, I resigned and started my own company.”
Aqueous moved into its current Rancho Cucamonga, CA, location six years ago, but the 15,000 sq. ft. facility still looks brand new. In January, CIRCUITS ASSEMBLY toured the facility with national sales manager John Hall, who has been with the company for nine years.

Figure 1. Aqueous Techologies’ current headquarters in Rancho Cucamonga, CA.
All around the facility, there is an extended family feel. “I started working on the assembly line in college just to pay for a trip to Ireland and Scotland,” Hall recalled. Today, Hall is not only one of the firm’s 30 staffers, 28 of whom work onsite, he is also Konrad’s son-in-law. Almost all of them are in production, and 80% of the building is designated for manufacturing.
Aqueous believes its single-minded approach is an advantage. “Our only focus is cleaning machines,” Hall said, giving Aqueous “the luxury of putting more thought into the machines” to become “more and more environmentally conscious.”
Perhaps that’s why Aqueous boasts the biggest market share in the US for batch machines, the only type of washer the company designs and builds. Given that the assembly market has trended away from inline equipment, competitors have taken notice. “Every inline manufacturer, without exception, has built a batch cleaner in the last few years,” asserts Hall.
The facility is a testament to the industry’s response. In one conference room stands a glass case that houses more than 40 industry awards (including multiple Circuits Assembly New Product Introduction Awards). Across the hall is a cleaning and analysis demo site for customer samples, where clients can “dial in” on what they need before buying a machine, Hall explains.
The demo room includes a cleaner, a tester and visual analysis. The company’s flagship Trident cleaner and defluxer is designed to remove all flux types from an assembly, according to Hall, and to do so in any mode of discharge: full, low or (the most popular) zero. Like its customers, Aqueous is “conscientious about how much water, power and chemicals we use and how we dispose of them,” says Hall.
The Trident machine reuses water and holds onto the chemicals. “Aqueous cleaners are designed not to go into the drain. Our zero-discharge systems filter and re-deionize solution and rinse water to alleviate draining requirements.
“Flux conducts electricity. We remove flux at the end so voltage only goes where you want it to go. Flux allows dendrites to grow, so we remove them,” said Hall. “How clean a board needs to be depends on where it’s going. How long does it have to be in the field?”
Besides the napkin, the site contains other nods to the past. Hall points out the Zero-Ion G3 cleanliness tester in the showroom, an updated version of the machine originally developed in the 1970s in the US Navy. Designed to indicate assembly cleanliness, the system also takes quick, accurate measurements on bare boards and other electromechanical devices. Aqueous still sells the machine today.
Also in the showroom are a microscope and camera. Residues that cannot be seen visually are subject to a ROSE (resistivity of solvent extract) tester. Roughly one in three boards is sent out to special labs such as Foresite in Indiana, ACI Technologies in Philadelphia, or one of Trace Labs’ sites for ion chromatography testing. The labs can determine “‘where and what’ is on the boards before we set up a cleaning process,” said Hall.
In the building’s main manufacturing area resides a raw stainless steel weld shop. After welding, a newly built machine is powder-coated to protect the frame.

Figure 2. All Aqueous’ machines start from a common frame and take about a month to build.
Aqueous builds all its machines from a common dishwasher frame purchased from an outside party (which Hall did not disclose). All the parts are removed from inside the frame, the most cost-effective option, and half are donated to a local engineering school. The dishwasher frames are “stamped stainless. There are no openings,” Hall says.
During the build process, parts are kitted and then inserted into the machine. Once it gets to the kitting stage, “from frame to chamber to everything in it” takes a couple days. The whole process of building a machine takes about a month. The Trident machine includes tanks, pumps, a blowing system, and resistivity probes, and it runs on a Windows touch-screen platform.
‘Designed for Anyone’
In practice, users first create a cleanliness profile, including cleanliness level, wash time, wash temperature, rinse time, dry time, dry temperature, chemical concentration, and then, since it is all preprogrammed, “the operator just has to hit start and stop.”
The manufacturing floor also contains an Eco-cycler machine, which, according to the company, “captures, filters, re-deionizes, and reuses rinse water from defluxing systems.” In short, it makes everything zero-discharge.
Hall says Aqueous embraces the so-called “dishwasher stigma.” The proof is industry acceptance, he points out. The type of equipment Aqueous builds has been legitimized because “every company has one,” he says.
Hall was visibly excited when asked about upcoming new products. At IPC Apex Expo in Las Vegas this month, Aqueous is introducing two machines that Hall says will be the “next answer” to trends in cleaners for years. Usually, Hall said, the company only introduces small changes to the inside of current machines that are good for the end-user. He looks forward to unveiling brand new equipment.
“Demand for cleaning is definitely ramping,” he said. The primary end-markets are consumer, which is starting to ramp more, and military/biomedical, which has always cleaned. Hall adds, “It all comes from no-clean fluxes. Sometime in the last decade, the consumer side went to no-clean fluxes,” which have a lower solids content.
‘More Failures’
With the move to smaller parts, there is “more failure because of dendrite growth. Component miniaturization and lead-free cause higher reflow profiles.” Hence the “giant ramp” in cleaning, he said. This is also because of increased use of conformal coating, which has found its way into all sorts of non Class 3 product.
Aqueous does a lot of telecom work with firms such as Qualcomm, L3 Communications and contractors that work with these companies. Cellphones, for instance, are coated to protect the microphone from moisture, which often comes from the user’s own mouth. As Hall says, “To be good for adhesion, the board has to be clean.”
“Cleaning is a three-step process,” he said, referring to chemicals, rinsing and drying. “With lower surface tension, it’s easier to get into small spaces.” All of Aqueous’ efforts lie in the second step: rinsing, which involves large pumps, spray systems and nozzles. A specific type of nozzle is used at a certain angle to push chemicals out from under components.
Asked if cleaning chemistries are designed for types of boards, fluxes or specific machines, Hall replied, “All of the above.”
Don’t expect drop-in chemistry, however. Chemicals are formulated for different types of metal, alloys, flux, coatings, plastics, inks and labels, Hall explained. “Some chemicals work better in batch or inline worlds,” and “certain cleaning chemistries tend to work better with certain machines.” For low-standoff components, for instance, Aqueous recommends a “focused mechanical energy,” said Hall.
To keep up with the changes in cleaning trends and, especially, chemistry, Aqueous has formed strong relationships with other firms in the industry, including Zestron, Kyzen and Petroferm. “We listen for manufacturing process trends and adjust equipment designs based on information from our industry partners.”
They also teamed with other companies in the industry to host 16 education-based cleaning workshops in the past year alone. More are being lined up for this year.
Reaching out to industry seems a natural extension of the culture here. At Aqueous, there is a real sense of friendly, down-to-earth teamwork, all in pursuit of Konrad’s green dreams. At home, Konrad might be Hall’s father-in-law, but in the office, Konrad is seen not just as a boss and mentor, but as a visionary.
Today, Aqueous Technologies has built more than 4,000 washers. Those built just last year have saved more than 36 million gallons of water, Konrad says. “We set out to build an unapologetic legitimate cleaning machine that was environmentally responsible. I believe in doing so, we have made the world a slightly better place.”
Chelsey Drysdale is senior editor for PCD&F/CIRCUITS ASSEMBLY; cdrysdale@upmediagroup.com.
A new CAD tool imports design files and quantitatively predicts product life.
Previous approaches to reliability assurance included “gut feel,” empirical predictions such as MIL-HDBK-217 and TR-332, industry specifications, “lessons learned” programs, failure mode effects analysis, and test-in reliability schemes. While these approaches can provide some value, it is felt that the most comprehensive analysis can best be done virtually using computer modeling of the circuit board in the intended environment. Such modeling can predict the life and reveal any design weaknesses before any prototype boards are even built.
The motivation for using modeling software lies in ensuring sufficient product reliability. This is critical because markets are lost and gained over reliability. Reputations can persist for years or decades, and hundreds of millions of dollars are at stake.
Using an automotive example, some common costs of failure:
- Total warranty costs range from $75 to $700 per car.
- Failure rates for E/E systems in vehicles range from 1 to 5% in first year of operation (Hansen Report, April 2005).
- Difficult to introduce drive-by-wire, other system-critical components.
- E/E issues will result in increase in “walk home” events.
Other Costs of Failure Examples
Type of Business Lost Revenue/ Hour
Retail brokerages $6,450,000
Credit card sales authorization $2,600,000
Home shopping channels $113,750
Catalog Sales Centers $90,000
Airline reservation centers $89,500
Cellular service activations $41,000
Package shipping services $28,250
Online network connect fees $22,250
ATM service fees $14,500
Supermarkets $10,000
The foundation of a reliable product is a robust design. A robust design provides margin, mitigates risk from defects, and satisfies the customer. Assessing and ensuring reliability during the design phase maximizes return on investment. The cost associated with finding a design flaw increases greatly the longer it takes to find it. Some have estimated this cost to be:
- Found during design: 1x.
- Found during engineering: 10x.
- Found during production: 100x.
- Found at customer: 1000x.
Electronics OEMs that use design analysis tools hit development costs 82% more frequently, average 66% fewer respins and save up to $26,000 in respins.1
MTTF / MTBF. Many companies use mean time to failure or mean time between failures calculations as their only means of assessing the reliability of their product while in the design stage. MTTF applies to non-repairable items, while MTBF applies to repairable items. They are based on the exponential distribution:
- Distribution: F(t) = 1 – e-λt
- Density (pdf): f(t) = λ e-λt
- Survival (sf): S(t) = e-λt
- Failure rate: λ(t) = f(t) / S(t) = λ e-λt / e-λt = λ
- MTTF: = 1 / λ (mean time to failure)
MTBF is typically calculated through a parts count method. Every part in the design is assigned a failure rate. This failure rate may change with temperature or electrical stress, but not with time. Failure rates are summed and then inverted to provide MTBF. Most calculations assume single point of failure, while some calculations take into consideration parallel paths.
A variety of handbooks provide failure rate numbers. These include MIL-HDBK-217, Telcordia, PRISM, 217Plus, RDF 2000, IEC TR 62380, NSWC Mechanical, Chinese 299B, HRD5. Some companies use internally generated numbers.
MTBF/MTTF calculations tend to assume that failures are random in nature and provide no motivation for failure avoidance. And, it is very easy to manipulate numbers with tweaks made to reach desired MTBF, such as modifying quality factors for each component. These calculations are also frequently misinterpreted. Example: A 50,000 hr. MTBF does not mean no failures in 50,000 hr.; it means half the products could fail by 50,000 hr. Basically, these calculations are a better fit toward logistics and procurement, not failure avoidance. Furthermore, these calculations take into account only failure of the components on the board, not wear-out mechanisms such as solder joint failures, plated through-hole fatigue, or damage due to vibration or shock events.
Design-build-test-fix approach. A common approach to product development is the design-build-test-fix (DBTF) approach. This is essentially a trial-and-error approach where the product is designed and prototypes are built. These prototypes then undergo testing; failures/defects are discovered; corrections are made in the design, and more prototypes are made, etc. Traditional OEMs spend almost 75% of product development costs on this approach.2 Shortcomings of this approach include:
- All design issues often not well-defined.
- Early build methods do not match final processes.
- Testing doesn’t equal actual customer’s use.
- Improving fault detection catches more problems, but causes more rework.
- Problems found too late for effective corrective action, fixes often used.
- Testing more parts and more/longer tests “seen as only way” to increase reliability.
- Cannot afford time or money to test to high reliability.
Recommended Design Approach
To design a product with the best chance for optimal reliability, one must understand the primary wear-out mechanisms and failure modes (often referred to as physics of failure). These wear-out modes can then be modeled and avoided with proper design improvements. One must also understand the expected life of the product and environment that the product will be exposed to over the course of its life. Finally, state-of-the-art tools should be used to understand the design and its impact on reliability before prototypes are built (while product is still in the design stage).
Electronics failures are typically attributed to a quality defect, overstress or wear-out. Quality defects often result from mistakes made in manufacture of the component or in PCB assembly. Design for manufacture and Six Sigma quality controls are required to minimize such defects and are outside the scope of this article. Overstress failures occur when a product is used outside its intended purpose or is exposed to an environment for which it was not intended (example, dropping your iPhone in the commode). This mode of failure is also outside the scope of this article. Avoiding wear-out failures in an electronics product is the focus since these failure modes can be modeled using established algorithms.
Wear-out failures have become much more common with aggressive design practices to shrink the footprint of electronics and design-in more applications. This has led to smaller, more closely spaced solder joints and through-hole vias. IC packages also have lower standoff and thus exert higher stress on solder joints during thermal cycle excursions. Additionally, lead-free solders have been introduced, which require higher assembly temperature and have different mechanical properties.
The advantage of wear-out failures is that we can understand how these failures take place and model them. For example, one researcher found that 65% of electronic failures were due to thermo-mechanical effects (CTE differences or diffusion).3 Common wear-out failure mechanisms that are modeled with Sherlock modeling software include:
- Thermal-cycle fatigue of solder joints.
- Mechanical vibration failure of solder joints.
- Shock failure of solder joints.
- Plated through-hole fracture.
Understanding Product Environment
Desired lifetime and product performance metrics must be identified and documented. The desired lifetime might be defined as the warranty period or by the expectations of the customer. Some companies set reliability goals based on survivability, which is often bounded by confidence levels such as 95% reliability with 90% confidence over 15 years. The advantages of using survivability are that it helps set bounds on test time and sample size and does not assume a failure rate behavior (decreasing, increasing, steady-state).
If the environment in which the product will be shipped and used is understood, then these conditions can be inserted in the model, and their impact on wear-out failure can be calculated. Mechanical vibration and shock, as well as thermal excursions, during shipping can be estimated depending on how the product is transported. Some products require long storage times or aggressive storage conditions, and these can also be modeled. Of greatest concern is how the product is used by the customer. One must estimate the number and magnitude of temperature excursions and mechanical stresses that the product will be exposed to while in use.
Defining environments. Several commonly used approaches are used to identify the environment. One approach involves the use of industry/military specifications such as MIL-STD-810, MIL-HDBK-310, SAE J1211, IPC-SM-785, Telcordia GR3108, and IEC 60721-3. Advantages of this approach include the low cost of the standards, their comprehensive nature and consensus industry agreement. If information is missing from a given industry, simply consider standards from other industries.
Disadvantages include the age of the standards; some are more than 20 years old, and lack validation against current usage. The standards both overestimate and underestimate reliability by an unknown margin (Figure 1).
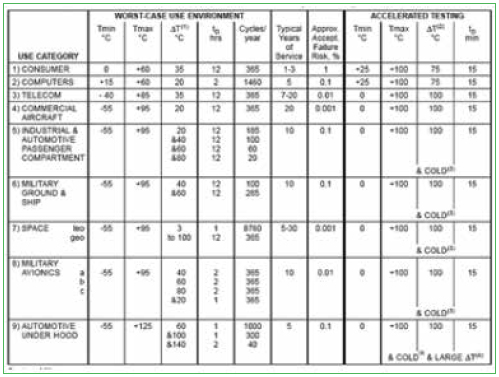
Figure 1. Some standards, such as IPC-SM-785, are too old to be inherently valid for today’s use.
Another approach to identifying the field environment is based on actual measurements of similar products in similar environments. This gives the ability to determine both average and realistic worst-case scenarios. All failure-inducing loads can be identified, and all environments, manufacturing, transportation, storage and field can be included.
In addition to thermal cycle environments, the reliability software accepts vibration and shock input as well. Figure 2 shows representation of this input. Identify the number of natural frequencies to look for within the desired frequency range. Single point or frequency sweep loading is available, and techniques are also available to equivalence random vibration to harmonic vibration.
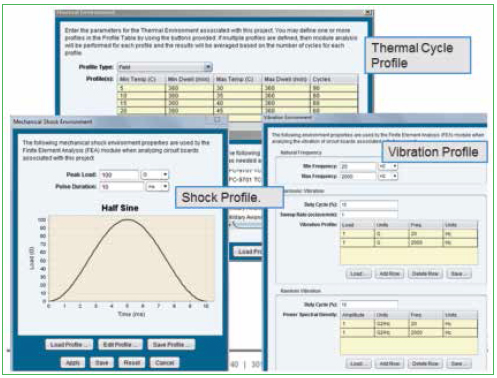
Figure 2. Environmental profiles inserted into software for modeling.
Vibration loads can be very complex and may consist of sinusoidal (g as function of frequency), random (g2/Hz as a function of frequency) and sine over/on random. Vibration loads can be multi-axis and damped or amplified depending on chassis/housing.
Transmissibility. The response of the electronics will be dependent on attachments and stiffeners. Peak loads can occur over a range of frequencies, including the standard range of 20 to 2000Hz and an ultrasonic cleaning range of 15 to 400kHz.
Vibration failures primarily occur when peak loads occur at similar frequencies as the natural frequency of the product or design. Some common natural frequencies:
- Larger boards, simply supported: 60 - 150Hz.
- Smaller boards, wedge locked: 200 - 500Hz.
- Gold wire bonds: 2kHz - 4kHz.
- Aluminum wire bonds: >10kHz.
Import files. The software is designed to accept ODB files, which contain all the data for the PCB, the components and their locations. Data can also be imported with Gerber files and an individual bill of materials (BoM). Figure 3 shows an example of a PCB stack-up and relevant data for reliability modeling.
Figure 3. PCB layer viewer and relevant data.
Parts list. Individual component data is part of the ODB file; however, modifications to the data can be made manually to ensure physical characteristics of all the components are accurate. Figure 4 shows the component editor, and Figure 5 shows the laminates and their properties that are embedded in the software.
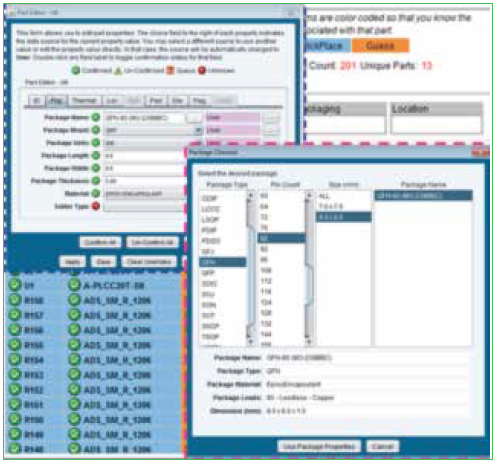
Figure 4. Parts list package database editor.
Analyses. Six analyses are currently conducted:
- CAF (conductive anodic filament) formation.
- PTH fatigue.
- Solder joint fatigue.
- Finite element simulations.
- Natural frequencies.
- Vibration fatigue.
- Mechanical shock.
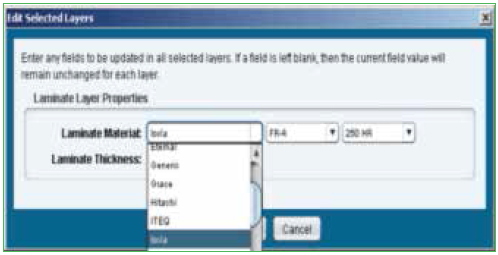
Figure 5. Laminate manufacturers.
CAF formation. Conductive anodic filament formation is when electrochemical migration of copper occurs between two barrel vias (Figure 6). The migration occurs through the PCB laminate and not on the surface (which is considered a different defect mechanism).
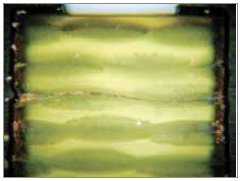
Figure 6. CAF formation between vias within the PCB.
One factor that drives CAF is damage to the laminate surrounding the drilled via. This can occur from a dull drill bit, excessive desmear etching or poorly laminated layers. Environmental factors that can increase the likelihood of CAF formation are the voltage across neighboring vias, spacing of the vias, and high temperature/humidity conditions. The software evaluates the edge-to-edge spacing of all the vias on the board and estimates the risk of CAF formation based on the damage around each via, as well as how well the product was qualified with CAF testing. Such vias can then be assessed to determine if there is a high voltage potential between them, or if they could be exposed to high humidity conditions.
PTH fatigue. PTH fatigue occurs when a PCB experiences thermal cycling. The expansion/contraction in the z-direction is much higher than that of the copper that makes up the barrel of the via. The glass fibers constrain the board in the x-y plane, but not through the thickness, so z-axis expansion can range from 40 to 70 ppm/°C. As a result, a great deal of stress can be built up in the copper via barrels, resulting in eventual cracking near the center of the barrel, as shown in the cross section photos in Figure 7.
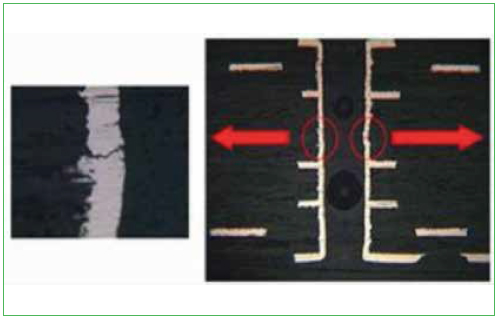
Figure 7. PTH fatigue images.
A validated industry failure model for PTH fatigue is available in IPC-TR-579, which is based on round-robin testing of 200,000 PTHs performed between 1986 and 1988. This model used hole diameters of 250µm to 500µm, board thicknesses of 0.75mm to 2.25mm and wall thicknesses of 20µm and 32µm. Advantages include the analytical nature in using a straightforward calculation that has been validated through testing.
Disadvantages include the lack of ownership and validation data that is approximately 20 years old. The model is unable to assess complex geometries, including PTH spacing and PTH pads that tend to extend lifetime. It is also difficult to assess the effect of multiple temperature cycles. However, this assessment can be performed using Miner’s Rule. The PTH equations take into account the expansion coefficient, PCB thickness, copper thickness, via diameter and glass transition temperature.
In addition to the series of algorithms used to calculate the fatigue life of PTHs, the quality of the copper plating is also taken into account. The “PTH Quality Factor” is a means of estimating the quality of the PTH fabrication process. This is a somewhat subjective determination. Rough edges of the copper wall will provide crack initiation sites and would reduce the quality. On the other hand, smooth copper walls, along with a surface finish such as ENIG, would improve the quality of the PTH. An example of a failure curve for PTH thermal cycle fatigue is shown in Figure 8, along with a list of vias in order of their expected life.
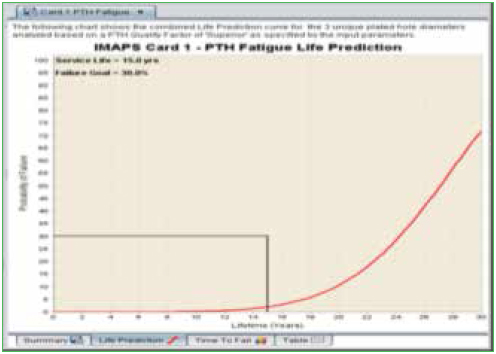
Figure 8. PTH fatigue life prediction.
Solder joint fatigue. Solder joint fatigue failures are becoming more prevalent due to the continued shrinkage of solder joint size and pitch that comes with more advanced packages. The reliability software takes into account the physical characteristics of the package and PCB to calculate thermal cycle fatigue life of the solder joints. The user may select eutectic tin-lead (SnPb), SAC 305 or SN100C (SnCuNiGe). Solder may be specified at the board or component level.
Solder Fatigue Model: Modified Engelmaier
The modified Engelmaier model is used within the software, which is a semi-empirical analytical approach using energy-based fatigue.
First, determine the strain range, Δγ, using:![]()
where C is a correction factor; LD is diagonal distance; a is CTE; ΔT is temperature cycle, and h is solder joint height. C is a function of activation energy, temperature and dwell time. LD is described further. Da is a2 – a1 and hs s defaults to 0.1016mm.
Next, determine the shear force applied to the solder joint using:![]()
where
F = shear force,
LD = length,
E = elastic modulus,
A = area,
h = thickness,
G = shear modulus, and
a = edge length of bond pad.
For the subscripts: 1 is the component; 2 is the board; s is the solder joint; c is the bond pad, and b is the board. This model takes into consideration foundation stiffness and both shear and axial loads. Leaded models include lead stiffness.
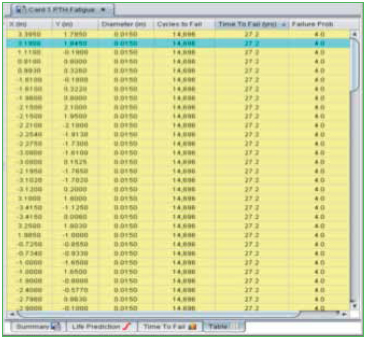
Figure 9. Tabular PTH fatigue life data.
Area. A1 is thickness of component (h1) x solder joint width, and A2 is thickness of board (h2) x solder joint width. As is length of solder joint (Ls) x solder joint width, which defaults to 45% of LD.
Ac is the length of the bond pad (Lc) x solder joint width. Lc defaults to 60% of LD.
Remaining parameters (h, G, v, a). Thickness: hs defaults to 0.1016mm, and hc defaults to 0.035mm.
Gs = Es / 2 x (1+vs) where Es = Temperature dependent modulus of solder and vs = 0.36.
Gc = Ec / 2 x (1 + vc) where Ec = 120 GPa and vc = 0.3.
Gb = Ec / 2 x (1 + vb) where Eb = 17 GPa and vb = 0.18.
a = √As.
Then, determine the strain energy dissipated by the solder joint using:
![]()
Calculate cycles-to-failure (N50), using energy-based fatigue models for SAC developed by Syed – Amkor:![]()
and using the energy-based model for SnPb
![]()
The software also has user overrides for solder fatigue.
Validation of Modeling Results
How accurate are the software’s modeling results compared with actual data? To answer this, more than 100 models were run with individual components and the results compared with reliability data from the literature. The results for QFNs, QFPs and BGAs are shown in Figure 10. The predicted results are on the x-axis and the modeled results on the y-axis. A perfect model would result in a diagonal line.
Figure 10. Predicted thermal cycle results compared with modeling results.
Naturally, there is variation in the results; however, for the most part, the predicted results are within a 10% band of the actual data. A larger
scatter in data is seen for BGAs, as is typical of experimental results for these components.
Unreliability. Thermal cycle results are provided as an unreliability plot that represents the cumulative reliability of all the components on the circuit card assembly (CCA) (Figure 11). The software will also show the rank order of individual components and their respective reliability so that the weakest links are determined (Figure 12). When a product consists of several CCAs, an unreliability failure plot is provided that takes into account all the assemblies.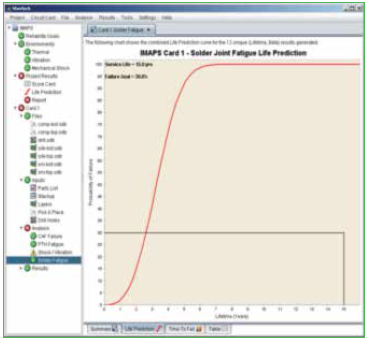
Figure 11. Solder joint fatigue life prediction (representing a very harsh under hood environment).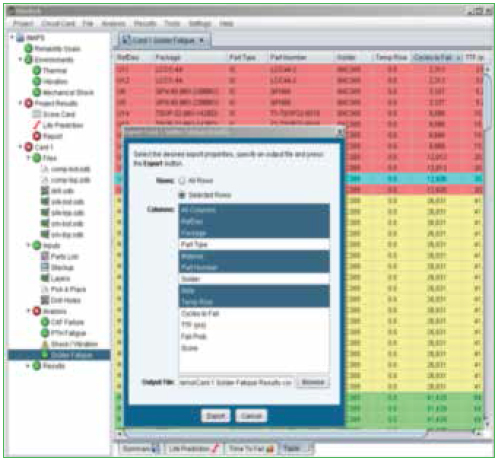
Figure 12. Sherlock results table.
Natural frequency analysis. The software contains an embedded finite element modeling tool that allows the user to select the mesh size and angle. The FEA is used to calculate the natural frequencies of the CCA, as well as the vibration and shock behavior. An example of the mesh created for a CCA is shown in Figure 13, followed by the first natural frequency generated for the assembly – based off the mount points used for the card.
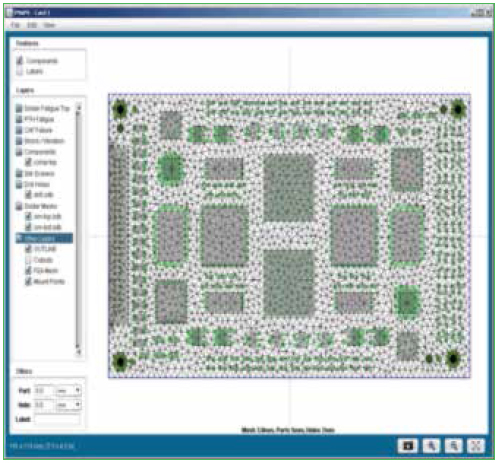
Figure 13. Mesh and mount points.
Vibration fatigue. Lifetime under mechanical cycling is divided into low cycle fatigue (LCF) and high cycle fatigue (HCF). LCF is driven by plastic strain and modeled by Coffin-Manson.
![]()
-0.5 < c < -0.7; 1.4 < -1/c > 2
HCF is driven by elastic strain and modeled by Basquin.![]()
-0.05 < b < -0.12; 8 > -1/b > 20
Vibration software implementation. The software uses the finite element results for board level strain in a modified Steinberg-like formula that substitutes the board level strain for deflection and computes cycles to failure. Critical strain for the component is defined by: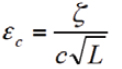
where
ζ is analogous to 0.00022B but modified for strain;
c is a component packaging constant (1 to 2.25), and
L is component length.
The Miles Equation relates Harmonic vibration to random vibration and must be utilized until the random vibration FEA code is fully tested and released.
![]()
where
fn = Natural frequency,
Q = transmissibility, and
ASDinput = Input spectral density in g2/Hz.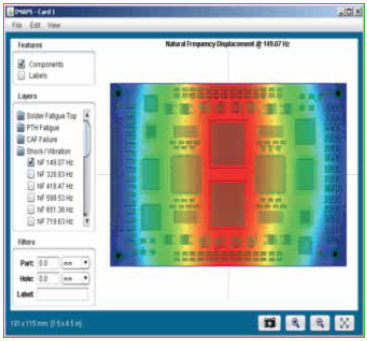
Figure 14. Natural frequency displacement.
The reliability software vibration modeling results show the displacement of the PCB at all locations (Figure 15). The results are plotted for each axis of vibration, and the most impacted components are revealed in the component list (Figure 16). Fatigue results are also shown in an unreliability plot over the life of the product, in the case where vibration is an ongoing event.
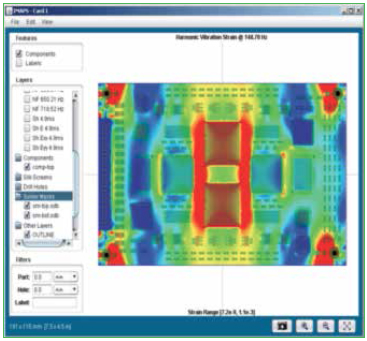
Figure 15. Graphical vibration results.
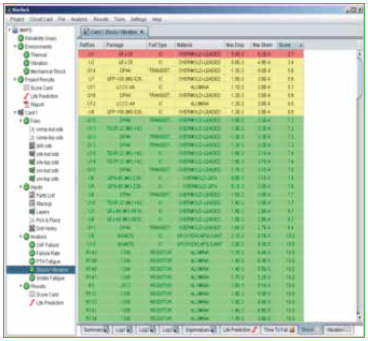
Figure 16. Graphical vibration results.
Mechanical Shock Environments
Mechanical shock requirements were initially driven by experiences during shipping and transportation. Shock became of increasing importance with the use of portable electronic devices and is a surprising concern for portable medical devices.
The basic environmental contributing factors include:
- Height or G levels.
- Surface (e.g., concrete).
- Orientation (corner or face; all orientations or worst-case).
- Number of drops.
JEDEC shock failure. Failures related to mechanical shock typically cause pad cratering (A,G in the image) and intermetallic fracture (B, F in Figure 17). This is an overstress failure, not a fatigue failure, and follows a random failure distribution.
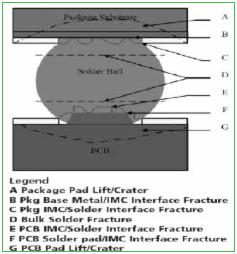
Figure 17. JESD22-B110A, Subassembly Mechanical Shock.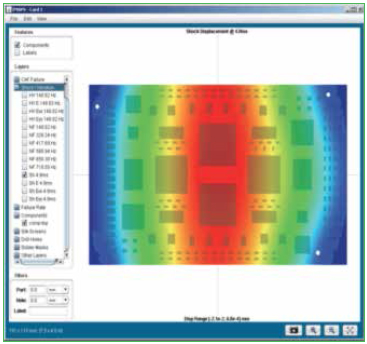
Figure 18. Shock displacement results for a test board.
The software analyzes shock based on a critical board level strain and will not predict how many drops to failure. Either the design is robust with regard to the expected shock environment, or it is not. Additional work has been initiated to investigate corner staking patterns and material influences. An example of the modeling showing displacement across a CCA after a shock event is shown in Figure 18. The rank order of components experiencing the largest strain is shown in Figure 19.
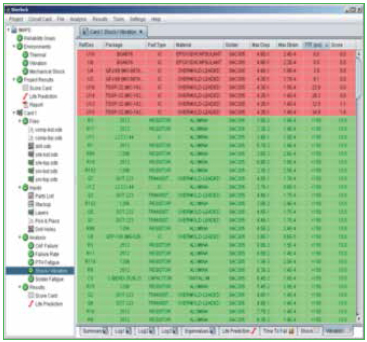
Figure 19. Components are listed in order of those experiencing the highest strain.
Constant failure rate module. A recent addition to the software has been the inclusion of a constant failure rate model using MIL-HNBK-217F calculations. Inputs necessary to compute failure rates are located in the parts list (Figure 20). The component failure rate is based off the 217F model and takes into account the temperatures at which the product operates. An example of the unreliability failure plot is shown in Figure 21, along with failure rates from solder joint fatigue and vibration.
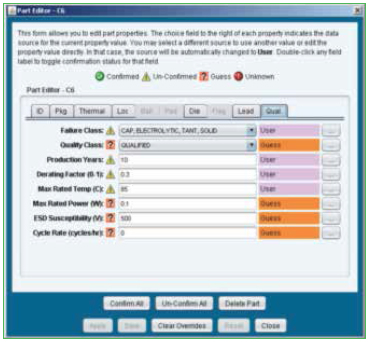
Figure 20. Failure rate information entry.
The appropriate test conditions can be determined by first generating a solder joint fatigue model based on the expected field conditions of the product. The percent failure at the required life is then known for the design. The model is then rerun using the desired thermal cycle test conditions (say 0° to 100°C). The number of cycles required to generate the same percent failure shown in the previous model is how many cycles are required (with no great percent failure). Naturally, the number of cycles may be increased if the sample size is reduced.
Early in the design phase of a product is the best time to run various what-if scenarios for the design. These might include experimenting to determine where the mount point locations should be in order to reduce strain on sensitive components. One may also run thermal cycling modeling using the various package options available for critical integrated circuits. The impact of changing a product from SnPb to Pb-free solder may also be evaluated.
Some high-reliability products require 100% ESS to ensure no poorly built products escape production. A common ESS test is thermal cycling; however, one does not wish to remove more than 5% of the useful life of the product during the ESS. By modeling the total life, it can be ensured that the number of cycles selected for ESS is appropriate.
Finally, many consumer electronics companies provide a warranty period for their products. Funds must be set aside for each product shipped to cover the expected field returns within the warranty period. It is important that these costs be roughly accurate and based on data, since money is lost if the retained amount is too large or too small. The results provided by the modeling tool can provide a portion of the total expected field returns due to hardware wear-out mechanisms.
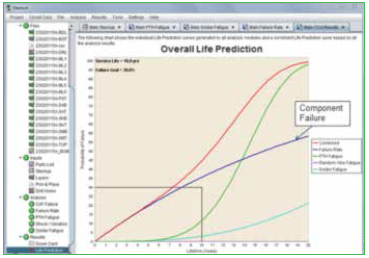
Figure 21. Life prediction that combines component failure with PTH and solder fatigue.
Summary
Designing in reliability upfront pays off immensely over the life of the product. To date, there has not been a simple-to-use method of estimating the wear-out life of an electronics product. The reliability software described here is designed to fill this need and does so by permitting rapid assessment of electronics systems reliability using physics of failure (PoF).
References
1. Aberdeen Group, “Printed Circuit Board Design Integrity: The Key to Successful PCB Development,” 2007.
2. Gene Allen and Rick Jarman, Collaborative R&D, John Wiley & Sons, 1999.
3. B. Wunderle and B. Michel, “Progress in Reliability Research in Micro and Nano Region,” Microelectronics and Reliability, vol. 46, no. 9-11, 2006.
Ed.: This article is adapted from a paper from SMTA International 2013 and is reprinted here with permission of the authors.
Dr. Randy Schueller is senior member of the technical staff at DfR Solutions (dfrsolutions.com); rschueller@dfrsolutions.com. Cheryl Tulkoff is senior member of the technical staff at DfR Solutions; ctulkoff@dfrsolutions.com.
Press Releases
- Distron Corporation Expands In-House Capabilities with Automated Conformal Coating for Complex PCB Assemblies
- Eastek International Corporation Appoints David Strabel as Vice President of Manufacturing
- SEMI Projects Double-Digit Growth in Global 300mm Fab Equipment Spending for 2026 and 2027
- Koh Young Announces Two Sales and Service Changes to Strengthen its Customers in the US and Canada


